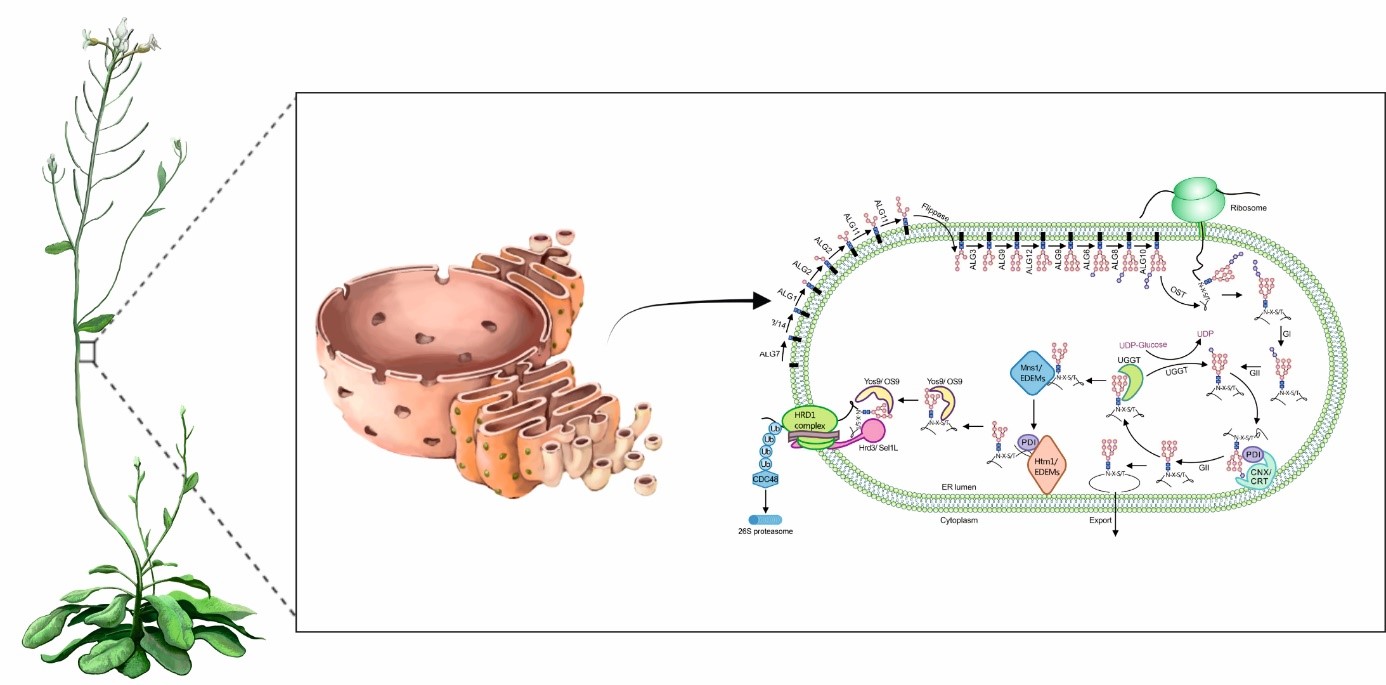
08.06.2022: Lisa THEISL (Graz Austria): Protein quality control and degradation in the endoplasmic reticulum, HS 02.11, Institut für Biologie, Bereich Zoologie, Universitätsplatz 2, 13:15 Uhr.
The endoplasmic reticulum (ER) is the entry gate for membrane and secretory proteins and serves as folding compartment. The native conformation of a protein is essential for its proper function, thereby protein folding is a crucial step in the maturation process of a protein. However, the folding process is error-prone. The ER is equipped with a highly-conserved quality control (QC) system to assist proteins to attain their native conformations. Several chaperones, lectins and enzymes of the ERQC are monitoring the folding process and enable the export of correctly folded proteins. Terminally mis-folded proteins are recognized by a multi-step degradation process, known as ER-associated degradation (ERAD). Most of our knowledge about ERQC and ERAD comes from studies in mammalian and yeast cells. Recently, it has been shown that plants are also equipped with homologous ERQC/ERAD components as well as unique plant-specific factors. However, the plant ERQC/ERAD system remains enigmatic, since many factors and mechanisms are still unknown. Possible strategies to discover novel components of the ERQC/ERAD system in the model plant Arabidopsis thaliana will be presented in this talk.